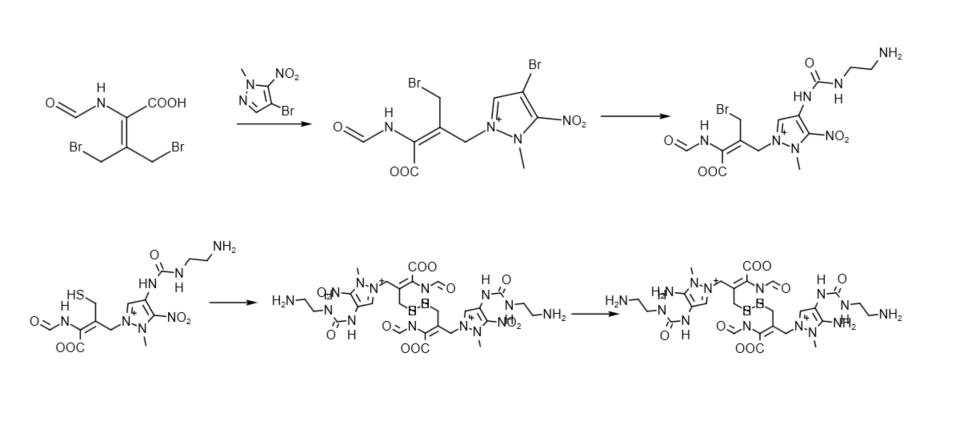
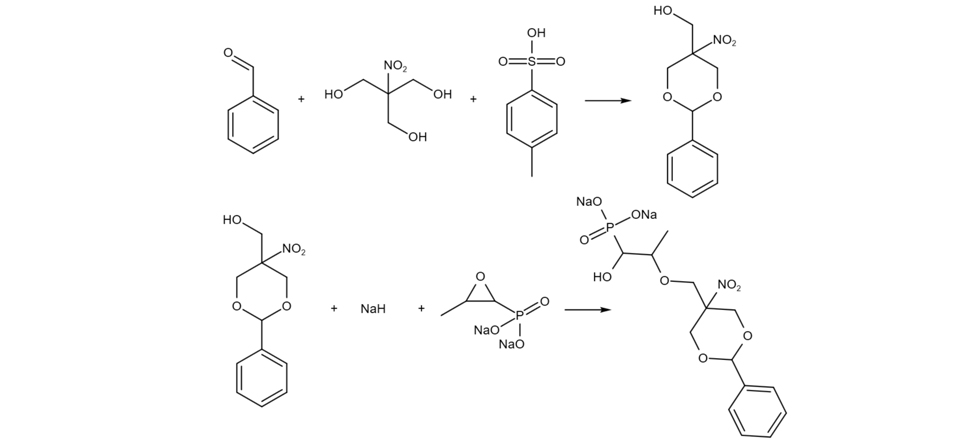
Intermediates Synthesis
Our expertise spans from drug impurity control to pharmaceutical intermediate synthesis, backed by decades of synthetic experience.
- Specializing in the synthesis of complex compounds
- The only ISO 17034 dual-system certified company worldwide
- A national high-tech enterprise
- A 4,000-square-meter R&D center
- A team of 70 domestic and foreign PhDs
- 150 R&D equipment, 120+ fume hoods
- 18 patents
Complete QMS / High-Quality Products & Services for Customers
Backed by a decade of innovation and certified to ISO 9001, ISO 17034, and CNAS, CATO maintains a perfect 100% audit pass rate with clients across the US, Australia, Poland, Japan, and leading Chinese pharma companies — enabling seamless, high-quality one-stop services for new drug development worldwide.
Over 150 sets (units) of equipment, ensuring quality assurance and reliability.
Quality is the cornerstone of CATO’s success. Guided by international standards, we employ advanced analytical tools — including GC-MS, NMR, and SEM — to ensure full control and traceability across all operations, from raw materials to delivery.
A 70+ PhD Research Team with Global Expertise

Preparation and Separation Laboratory
We develop chiral chromatography separation and preparation technologies for compound purification, and build structurally diverse chiral compound libraries through innovations in organic synthesis.

Process route research laboratory
We advance process development from lab research to pilot and intermediate-scale production, controlling trace impurities, improving yield, and optimizing reaction conditions for industrial-scale manufacturing.

Kilogram-scale laboratory
A versatile small-scale drug preparation platform for non-sterile intermediates, supporting product validation, pilot-scale production, and meeting customized requirements such as particle size, crystal form, residual solvents, and solubility.

Intermediate Synthesis Laboratory
Capable of conducting reactions under complex conditions with real-time structural elucidation, supported by ¹H NMR, ¹³C NMR, ¹⁹F NMR, and various 2D NMR techniques to ensure accurate structural confirmation of multi-chiral-center compounds.
1000+ Successful Delivery Cases Expertise in Synthesizing High-Difficulty Compounds
CATO serves over 500 pharmaceutical and biotech companies globally, delivering projects across the full development spectrum — from preclinical to commercialization. As of 2025, we have completed over 1,000 projects. Our core expertise includes advanced fluorine chemistry, asymmetric chiral synthesis, glycoside & biocatalysis technologies, and continuous-flow microreactor systems.
4000 m² Synthetic Chemistry Laboratory





Technical cooperation platform
While conducting independent research and development, the company has also established close cooperation with renowned institutions both at home and abroad. By fully leveraging the interactive advantages of production, education and research, the company has enhanced its research and development efficiency. At the same time, it has significantly accelerated the speed and intensity of its technological innovation, providing strong technical support for the company's development.