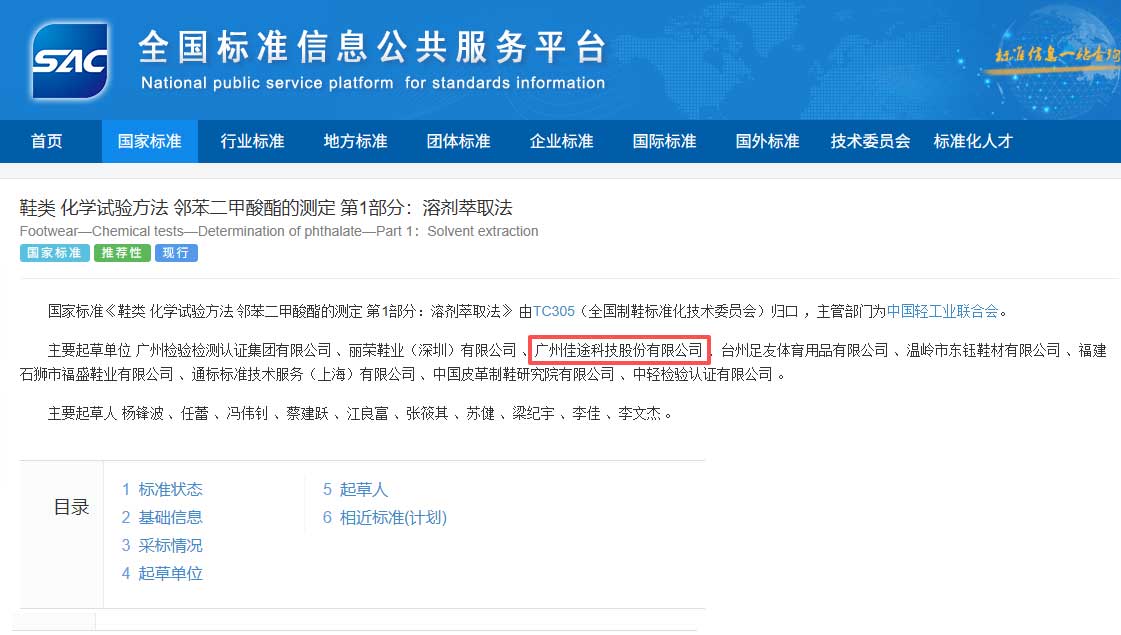
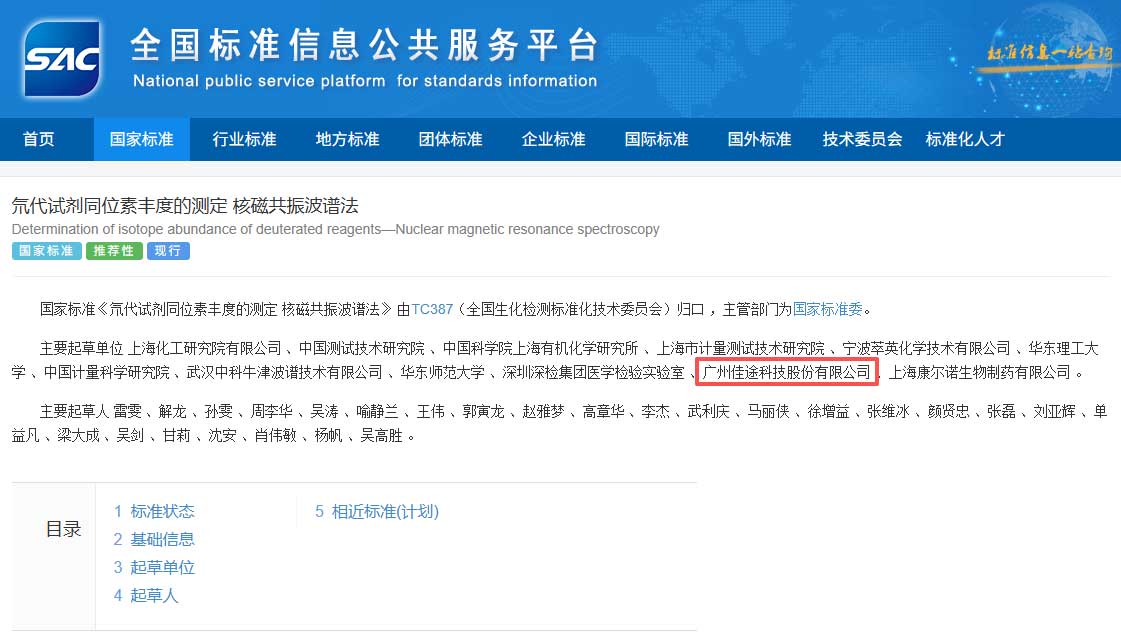
CATO Research Chemicals Inc. (Chinese corporate name: 广州佳途科技股份有限公司) was established in Guangzhou, China in November 2016. CATO is a technology-driven, vertically integrated manufacturer of certified reference materials (CRMs), analytical reference standards, and pharmaceutical impurity standards, sold under the CATO® brand.
Guided by the mission Make the Tests More Accurate, CATO serves a broad client base across the life sciences and chemical industries. Customers include pharmaceutical manufacturers, API and specialty chemical producers, CRO and bioanalytical laboratories, IVD and biotechnology companies, third-party testing organizations, regulatory and pharmacopoeial agencies, academic and research institutions, government bodies, hospital pharmacy departments, and advanced materials developers.
CATO's core services include: full-spectrum reference standards and CRMs, specialty chemical intermediates, custom and contract synthesis, pharmaceutical impurity isolation and structural elucidation, and analytical testing services.