REACH Annex XVII Adds New Restrictions on DMAC and NEP
On June 3, 2025, the European Union officially published Commission Regulation (EU) 2025/1090 in its Official Journal, introducing significant amendments to REACH Regulation Annex XVII by adding Articles 80 and 81. Two important industrial solvents—N,N-dimethylacetamide (DMAC) and 1-ethylpyrrolidin-2-one (NEP)—have been formally included in the restricted substances list.
On June 3, 2025, the European Union officially published Commission Regulation (EU) 2025/1090 in its Official Journal, introducing significant amendments to REACH Regulation Annex XVII by adding Articles 80 and 81. Two important industrial solvents—N,N-dimethylacetamide (DMAC) and 1-ethylpyrrolidin-2-one (NEP)—have been formally included in the restricted substances list.
Why Restrict These Two Substances?
Both DMAC and NEP are dipolar aprotic solvents characterized by high dielectric constants and high dipole moments. More importantly, both substances are classified by the EU as Category 1B reproductive toxicants, potentially causing harm to unborn children, thus making them priority chemicals for regulatory control.
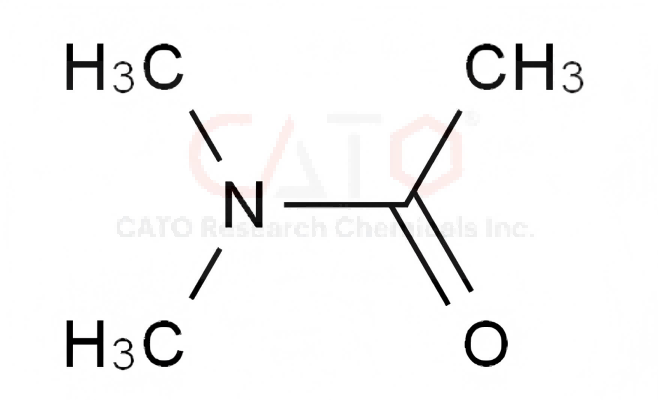
N,N-dimethylacetamide (DMAC)
CAS No. 127-19-5
EC No. 204-826-4
Catalog No.: LCEM501391-WA
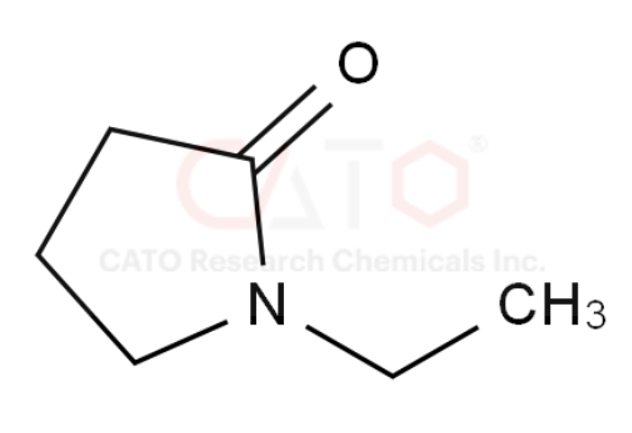
1-ethylpyrrolidin-2-one (NEP)
CAS No. 2687-91-4
EC No. 220-250-6
Catalog No.: CCHM702206
CATO Reference Standards Support Regulatory Compliance
With DMAC and NEP restriction regulations approaching full implementation, the compliance window for companies is rapidly narrowing. Establishing reliable analytical capabilities is not only a regulatory requirement but also a crucial guarantee for continued business operations.
CATO provides high-quality N,N-dimethylacetamide (DMAC) and 1-ethylpyrrolidin-2-one (NEP) reference standards, offering reliable analytical support to help companies successfully navigate regulatory challenges.



